|
10 Nonetheless, it is only in the last few years that the synthetic chemistry community has rediscovered this powerful tool for small scale synthetic applications. 4,6,15 In addition, electrochemistry can offer new reaction pathways that are complimentary to traditional synthetic chemistry procedures and can be particularly interesting for late-stage functionalisation due to its mild and selective nature. It therefore avoids the use of other harsh and often toxic chemical reducing and oxidising agents. 18,19 Electrochemistry offers a very mild and atom efficient method to achieve selective oxidative or reductive transformations using electrons as reactants. 1–17 This is exemplified by whole journal issues being dedicated to synthetic electrochemistry. Introduction During the last decade, electrochemistry has received renewed interest as a tool for synthetic chemistry. Current research includes electrochemical synthetic methods to metal compounds, flow technology and catalytic and biomedical applications. Charlotte was awarded a Royal Society Dorothy Hodgkin Fellowship which she took to Leeds in 2009. She completed a postdoctoral position with Prof Jonathan Steed in Durham, working on MOFs and metal-N-heterocyclic carbenes. She completed her PhD at the University of York working with Prof Francesca Kerton and Dr Jason Lynam on lanthanide catalysis and organophosphorus cages. He is currently working in the lab of Dr Charlotte Willans as a postdoctoral research assistant investigating the electrochemical synthesis of inorganic and organometallic complexes and the development of catalytic methodologies using high throughput experimentation.ĭr Charlotte Willans obtained her MChem from the University of York, conducting a research project at DSM (Netherlands) with Profs Johannes de Vries and André de Vries on palladium-catalysed cross-coupling reactions. Christiane is currently working in the group of Dr Charlotte Willans (University of Leeds, UK) as a postdoctoral research assistant on the development of a multifunctional electrochemical flow platform for high-throughput synthesis and optimisation of catalysts.ĭr Thomas Nicholls obtained his PhD in chemistry from the University of Tasmania in 2018 in the lab of Dr Alex Bissember, with research focused on the photophysical and electrochemical characterisation of novel copper-based visible light photocatalysts, and the development of photocatalysed synthetic methodologies enabling the rapid construction of complex organic frameworks. She then was awarded an RSC mobility grant to work with Dr Timothy Noël at Eindhoven University of Technology (Netherlands) on continuous electrochemical synthesis. During her PhD at Cardiff University (UK), Christiane worked with Dr Duncan Browne on multistep continuous flow processes using diazonium salts. We will discuss mechanistic considerations, the setup of an electrochemical reaction with all its components, trouble shooting and selected examples from the literature.ĭr Christiane Schotten graduated with a BSc and MSc from RWTH Aachen University (Germany). In this tutorial we will bridge the knowledge gap by providing an easily accessible introduction which will enable synthetic chemists new to the field to explore electrochemistry. This has led to a knowledge gap between experienced electrochemists and those new in the field. Despite the renewed interest there remains a barrier to widespread adoption of this technology derived from the extra knowledge and specialised equipment required. As such, encouraging broader application of electrochemistry by synthetic chemists should be a priority. Electrochemical synthesis offers a mild, green and atom efficient route to interesting and useful molecules, thus avoiding harsh chemical oxidising and reducing agents used in traditional synthetic methods. Zinc dissolves into the acid solution.A significantly renewed interest in synthetic electrochemistry is apparent in the increasing number of publications over the last few years. 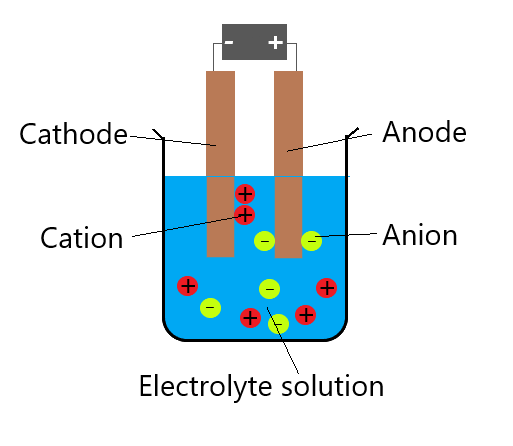 
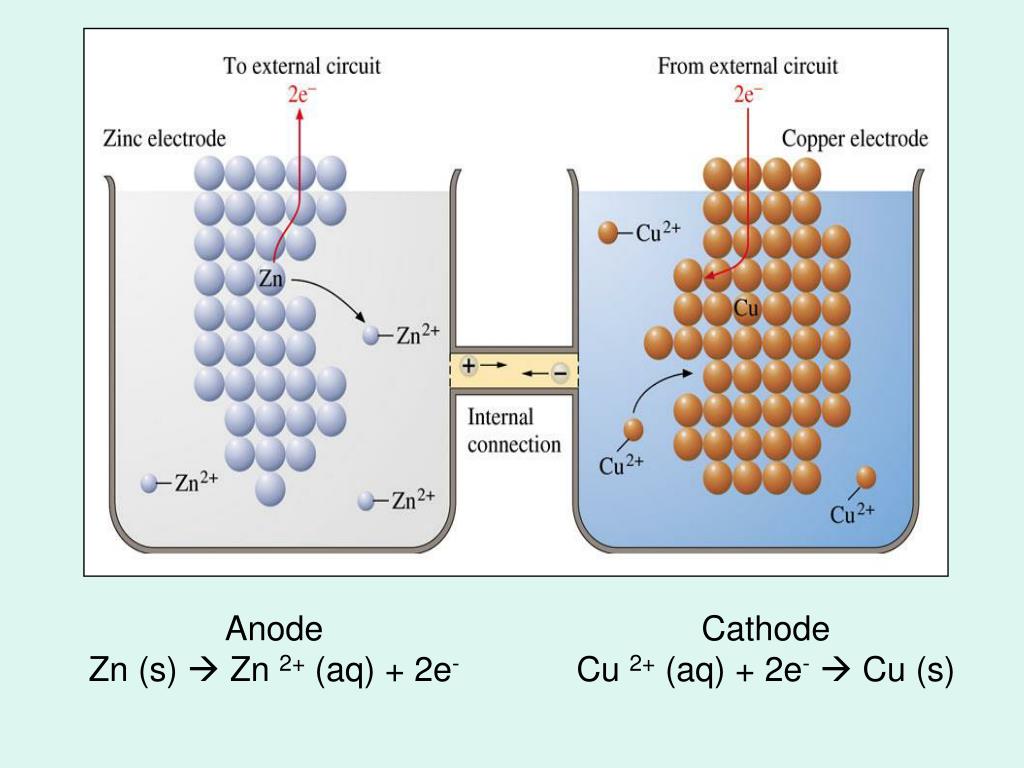
Each zinc atom loses two electrons and is oxidised to a zinc ion. Since zinc is more reactive than copper, oxidation will occur at the zinc electrode. The further apart the metals are in the reactivity series, the bigger the voltage of the cell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
